REAL WORK. REAL BARRIERS. REAL RESULTS.
Every engagement starts the same way: we go to the Gemba, find what's blocking the team, and remove it. Here's what that looks like in practice.
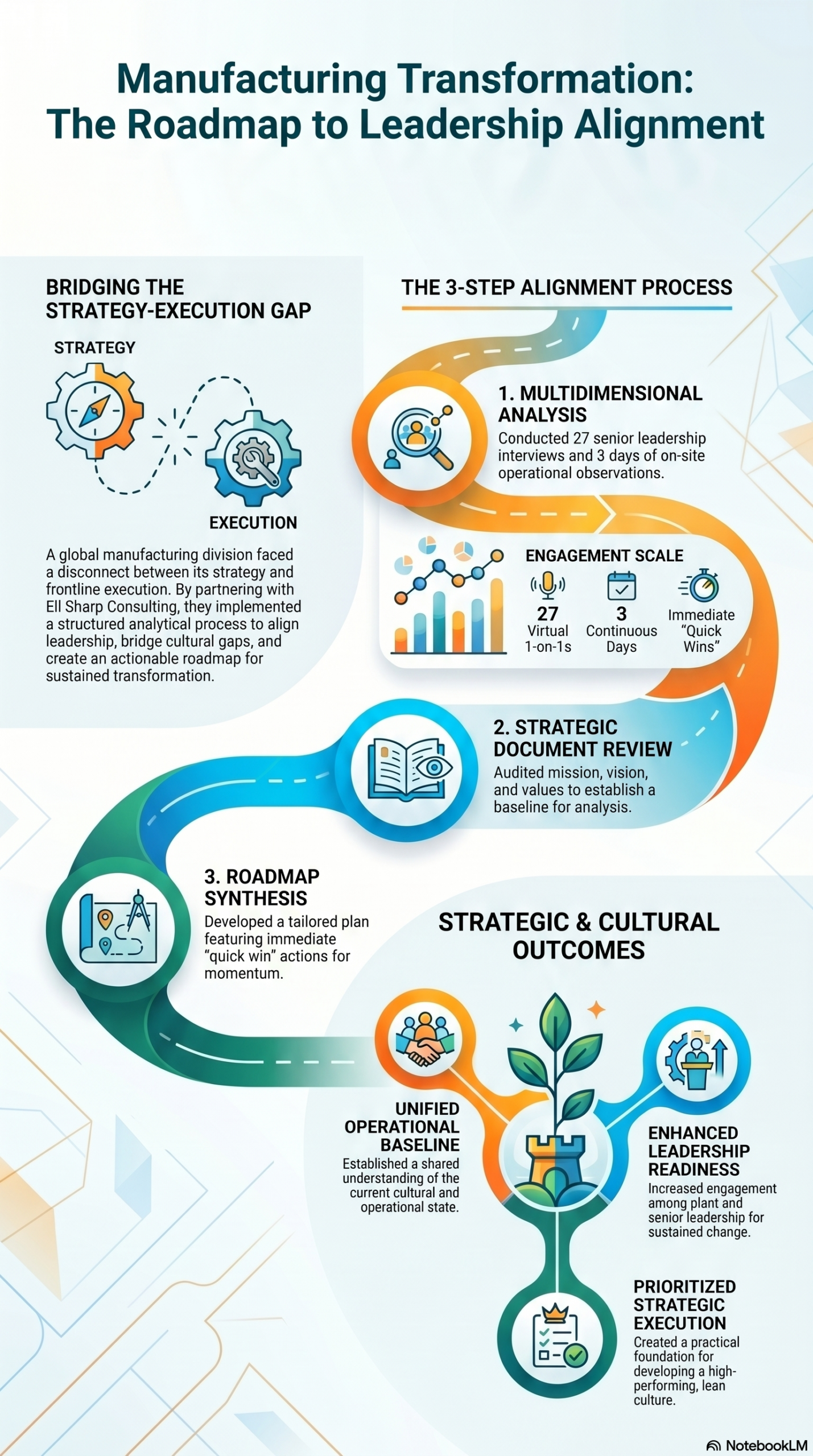
The plant that had 'always been this way'
THE SITUATION
A manufacturing division of a global corporation had a problem that everyone knew about and nobody had been able to fix: the gap between what leadership believed was happening on the floor and what was actually happening. Strategy wasn't connecting to execution. Cultural dynamics varied wildly between the plant and corporate teams. Improvement efforts kept stalling. The phrase leadership kept using — almost as a resignation — was that things had 'always been this way.'
Twenty-seven people across senior and plant leadership teams were interviewed. What emerged wasn't a skills problem. It was a systems problem — unclear priorities cascading into confusion, siloed decisions, and a leadership team that had never been given the tools to solve problems together. The people were capable. The system wasn't letting them move.
WHAT WE DID
We went on-site for three days — not to present recommendations, but to see. We attended meetings, walked the floor, observed how decisions actually got made. Then we worked with leadership to build a strategic roadmap grounded in what we'd seen together, not in what anyone assumed was true. The roadmap included immediate quick wins alongside longer-term alignment work, and was presented back to stakeholders in a way that created shared ownership rather than top-down instruction.
WHAT CHANGED
✓ Leadership left with a clear, actionable roadmap tied to their actual mission and values — not a generic framework
✓ Prioritized next steps gave the plant team immediate momentum rather than another planning cycle
✓ Senior and plant leadership began practicing the collaborative skills needed for sustained transformation — during the engagement, not after it
✓ The organization moved from 'this is just how it is' to a practical foundation for building a lean, high-performing culture
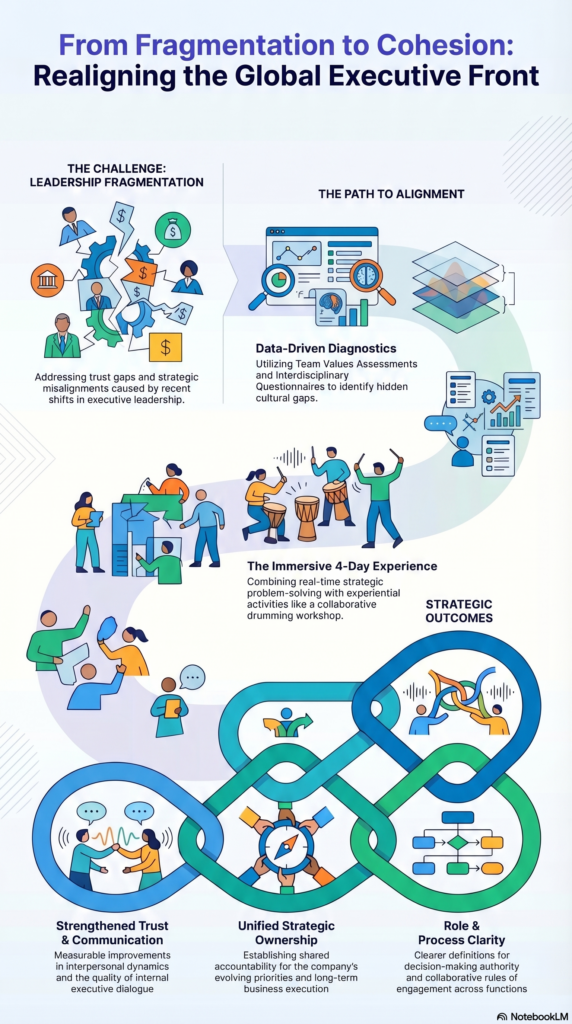
A leadership team that needed to learn to trust each other — fast
THE SITUATION
A global supply chain executive team at a healthcare company was going through a difficult transition: new leader, recent team member changes, shifting strategic priorities, and the urgent need to collaborate across functions that had historically operated independently. The team recognized the opportunity — and the risk. If they couldn't build real trust and alignment quickly, the next phase of strategic execution would stall before it started.
The challenge wasn't competence. Every person on that team was highly capable. The problem was that they hadn't yet built the shared language, relational trust, or collaborative habits to function as a unit under pressure. And in a global supply chain role, pressure is constant.
WHAT WE DID
We started with diagnostics — a Team Values Assessment and an Interdisciplinary Team Questionnaire — to surface the real gaps before designing anything. What we found shaped a four-day off-site workshop in France built around the team's live strategic challenges, not hypothetical exercises. They worked through real business problems together, designed their own collaboration processes, and established clear rules of engagement. On the third evening, a drumming workshop gave the team a completely different experience of what it feels like to create something together.
WHAT CHANGED
✓ Trust and communication improved measurably within the group
✓ The team built shared ownership of strategic priorities — not just awareness of them
✓ Cross-functional problem-solving became a practiced skill, not an aspiration
✓ Roles, responsibilities, and decision-making became clear
✓ The team left with the norms and processes to keep collaborating effectively — without needing another intervention


Zero late reports to the FDA — achieved in four days
THE SITUATION
A global MedTech company was submitting Medical Device Reports to the FDA late. Not occasionally — consistently. In a regulated industry where late MDR submissions carry real compliance risk, this was serious. Leadership knew it needed to change but previous efforts hadn't stuck. The process was fragmented, roles were unclear, and the team had never had a clear shared view of how work actually flowed from trigger to submission.
The deeper problem, as it often is, wasn't the people. It was that nobody had ever mapped the real process — what actually happened versus what was supposed to happen — with everyone in the room at the same time.
WHAT WE DID
We ran a four-day Lean Six Sigma Blitz with a cross-functional team: process users, subject matter experts, and representatives from connected departments. Day one was about seeing reality clearly — mapping what was actually happening and finding exactly where submissions were getting stuck. Day two the team redesigned the process together, with full accountability for what they were creating. Day three turned the redesign into a concrete action plan with named owners. Day four was a stakeholder presentation where leadership committed — publicly, in the room — to driving post-blitz actions to completion. We stayed on remotely after the engagement to monitor progress and help the team hold the gains.
WHAT CHANGED
✓ Zero late MDR reports — the target was met and sustained
✓ A global To-Be process was documented and implemented, covering both FDA and CAPA requirements
✓ The cross-functional team had clear roles and responsibilities for the first time
✓ Leadership was aligned and accountable — not just informed
✓ The team built the capability to prevent future lapses independently
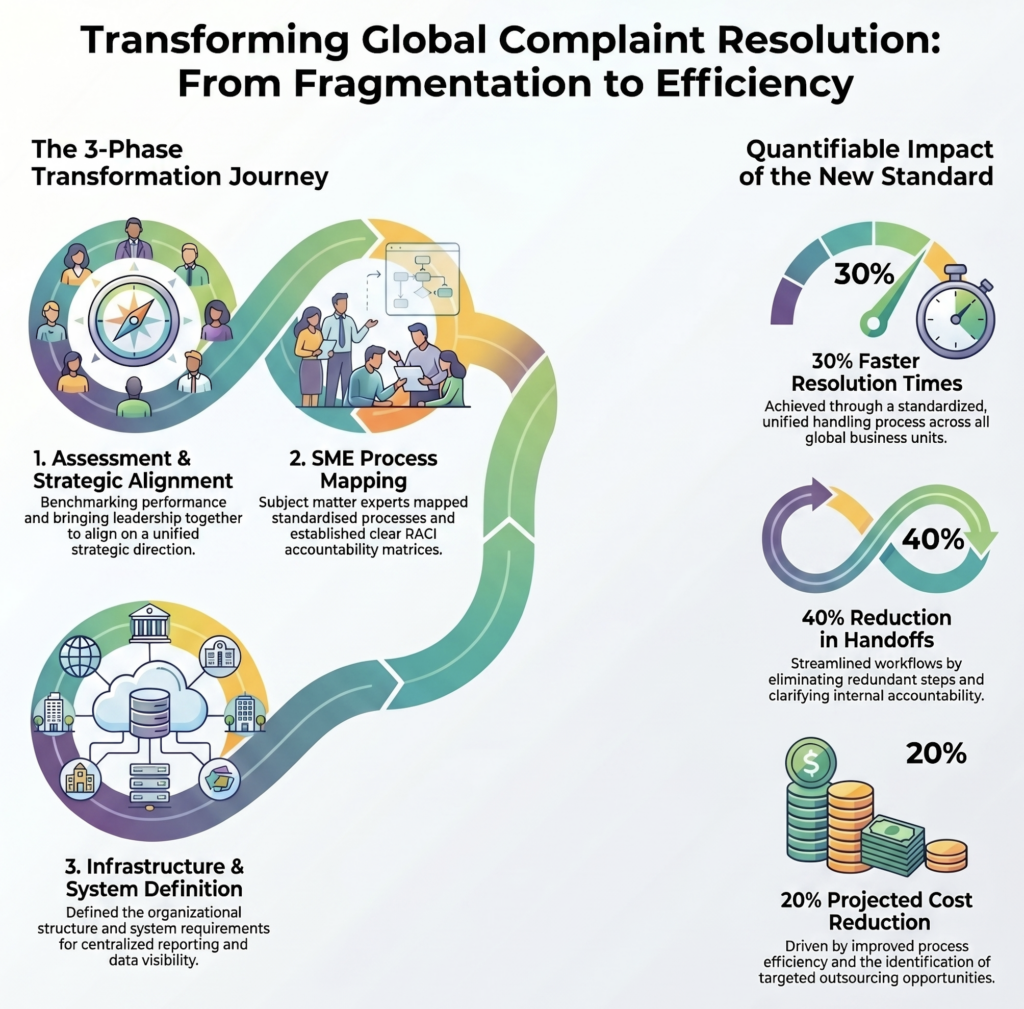
Complaint handling transformed across a global organization — 30% faster resolution
A global organization with complaint handling processes that varied by business unit — creating inconsistent response times, unclear accountability, and elevated costs.
Top result: Response times reduced by 30%. Handoff points cut by 40%. A centralized complaints function with a standardized process and clear accountability.
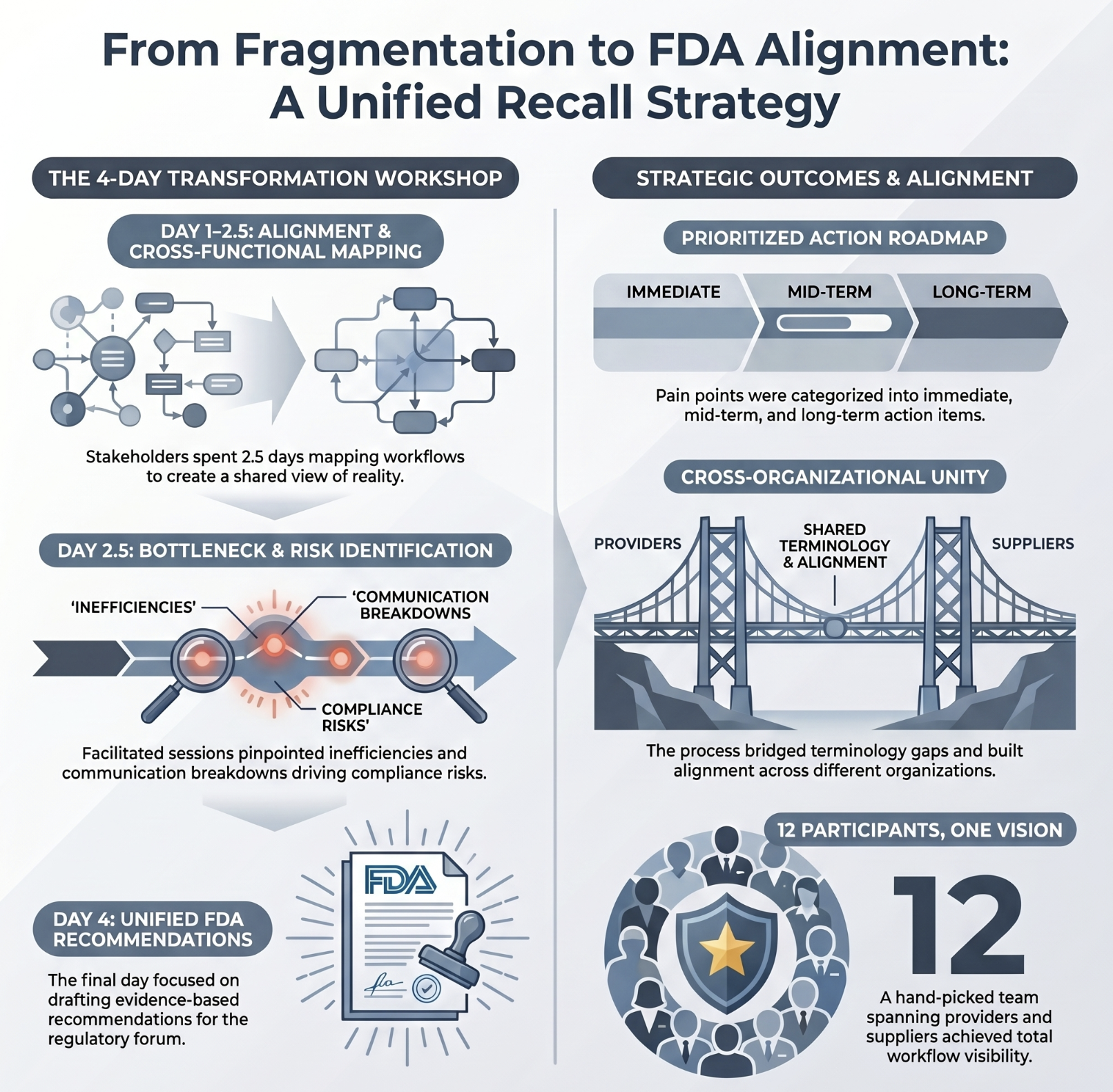
Recall process mapped — findings presented directly to the FDA
A consortium of healthcare providers and suppliers with fragmented, inconsistent recall processes and increasing regulatory scrutiny from the FDA.
Top result: Comprehensive process maps across both provider and supplier workflows. A prioritized improvement plan. A structured presentation delivered to the FDA.
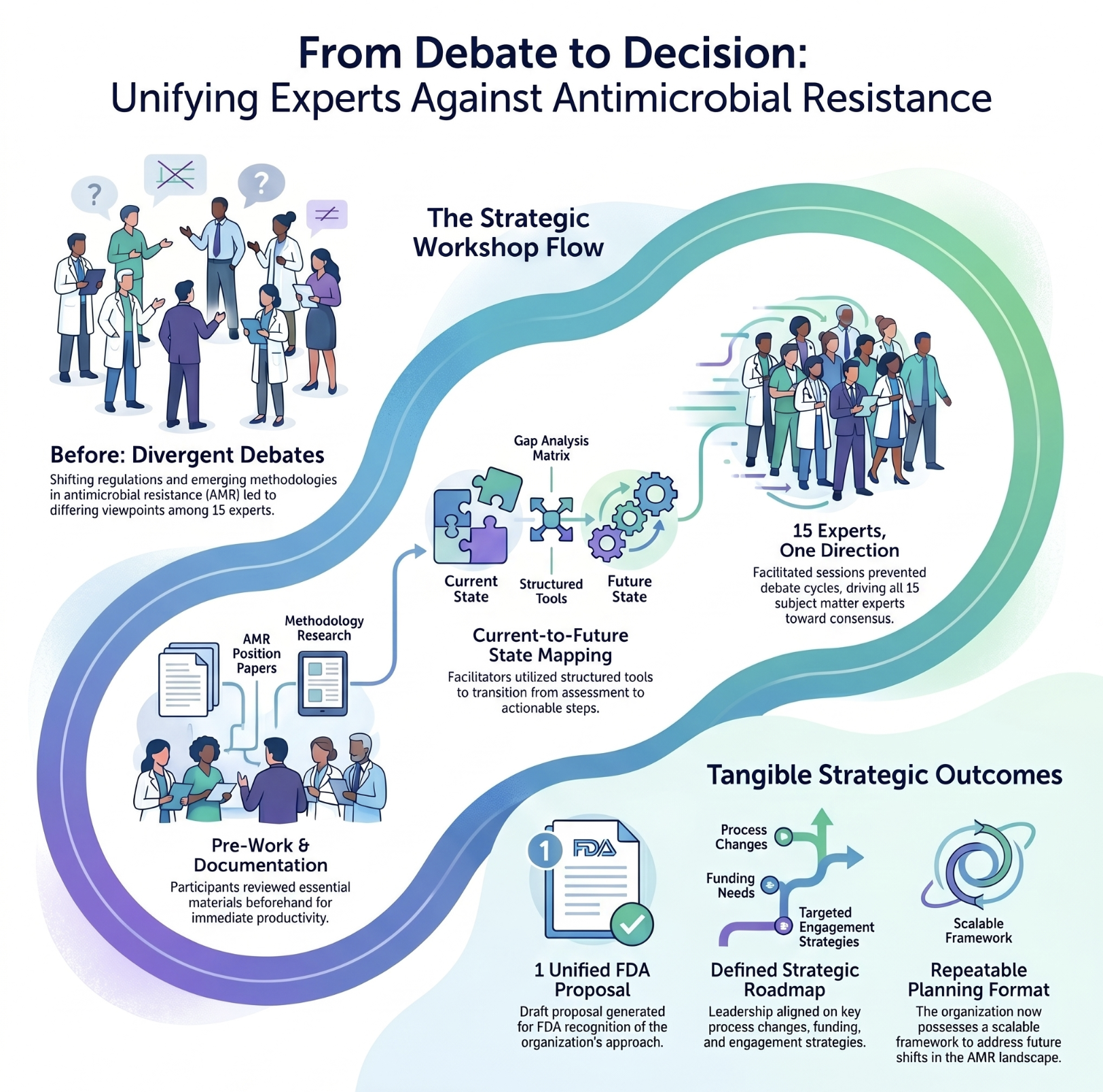
A standards organization finds its path forward on antimicrobial resistance
Fifteen subject matter experts needed to align on a unified strategy for breakpoints determination in a rapidly shifting regulatory landscape.
Top result: A prioritized action plan and FDA engagement strategy — built in a single day, with full expert consensus.

A new Operational Excellence team gets aligned — and stays that way
A recently restructured OE team with ambitious goals, varied backgrounds, and low relational trust — and a mandate they couldn't deliver on alone.
Top result: Shared strategic priorities, clear collaboration structures, and a team ready to drive results across the business.

Document development cycle time cut by 60% across 64 committees
A clinical standards organization with a document development process that had resisted previous improvement efforts — running across 64 committees.
Top result: 60% reduction in document development cycle time. A standardized process rolled out across all 64 Document Development Committees.
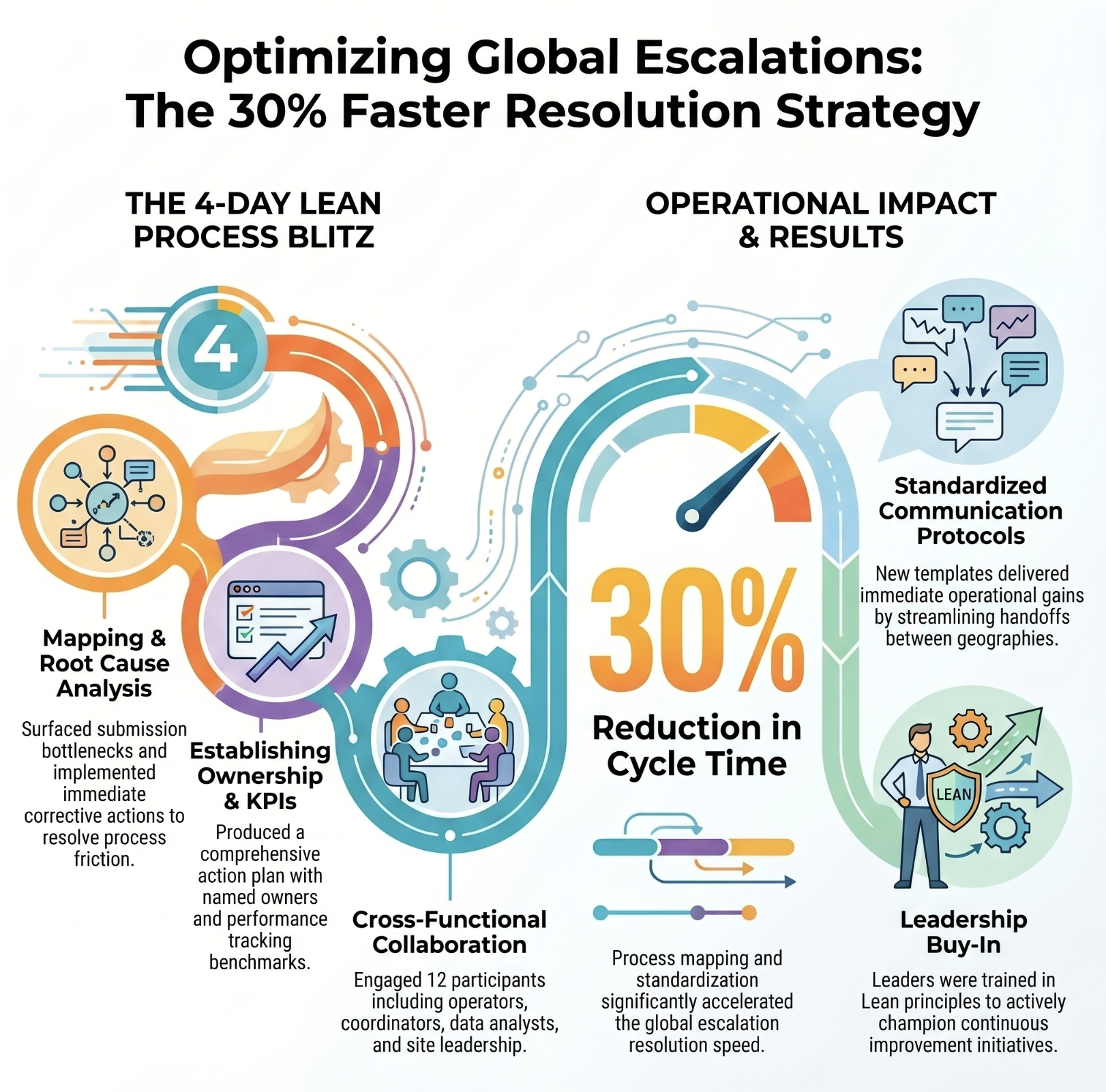
Global escalation process streamlined — 30% faster resolution
A global contact lens manufacturer with an escalation process causing delays, communication breakdowns, and compliance risk.
Top result: Escalation cycle time reduced by 30%. Leadership equipped and aligned to sustain the improvement.
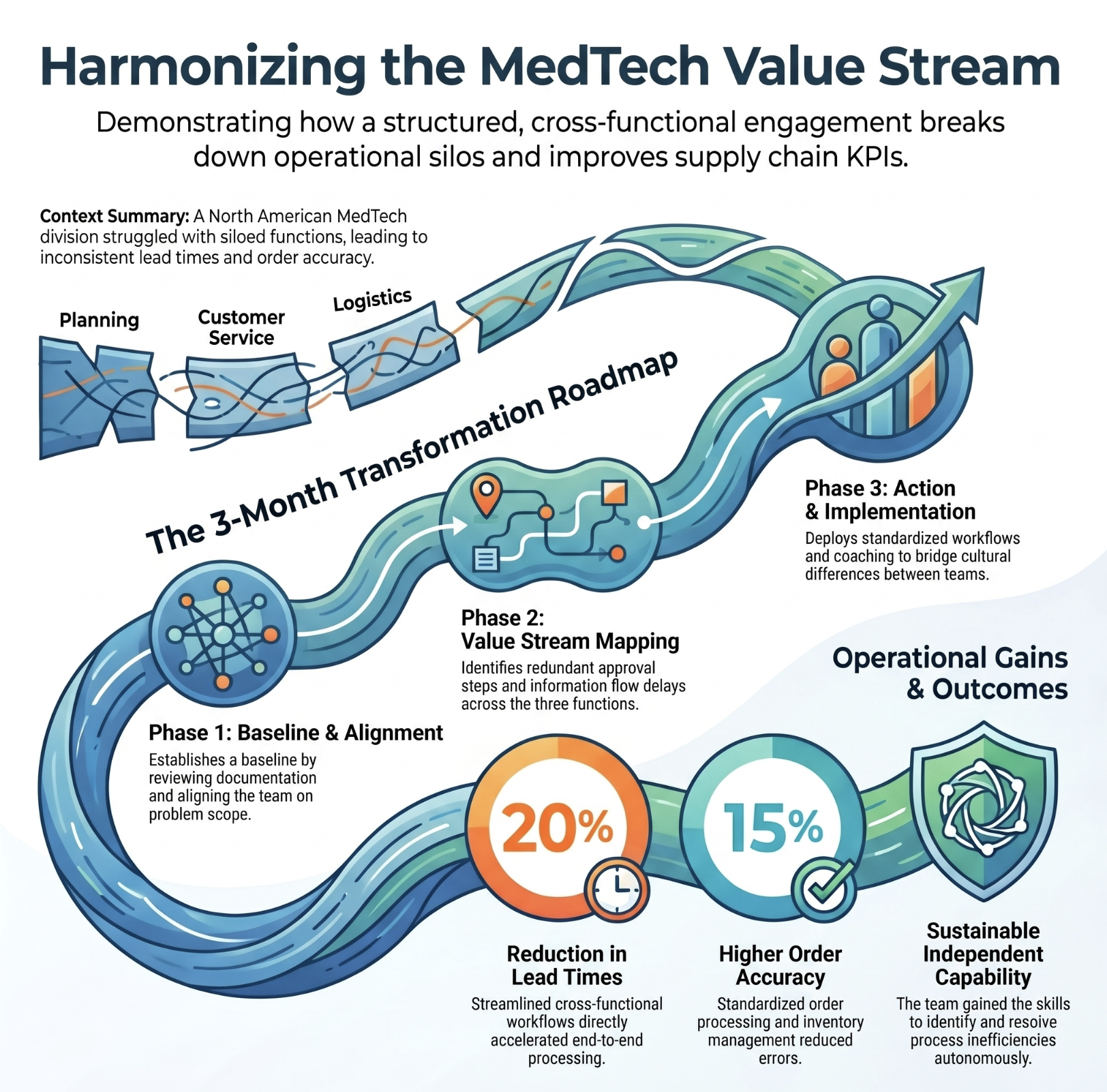
North American operations harmonized across Planning, Customer Service and Logistics
Three functions working independently with inconsistent processes, leading to inefficiencies and a lack of cohesion across a North American division.
Top result: Lead times reduced by 20%. Order accuracy improved by 15%. A cross-functional team now measuring and improving their own process.
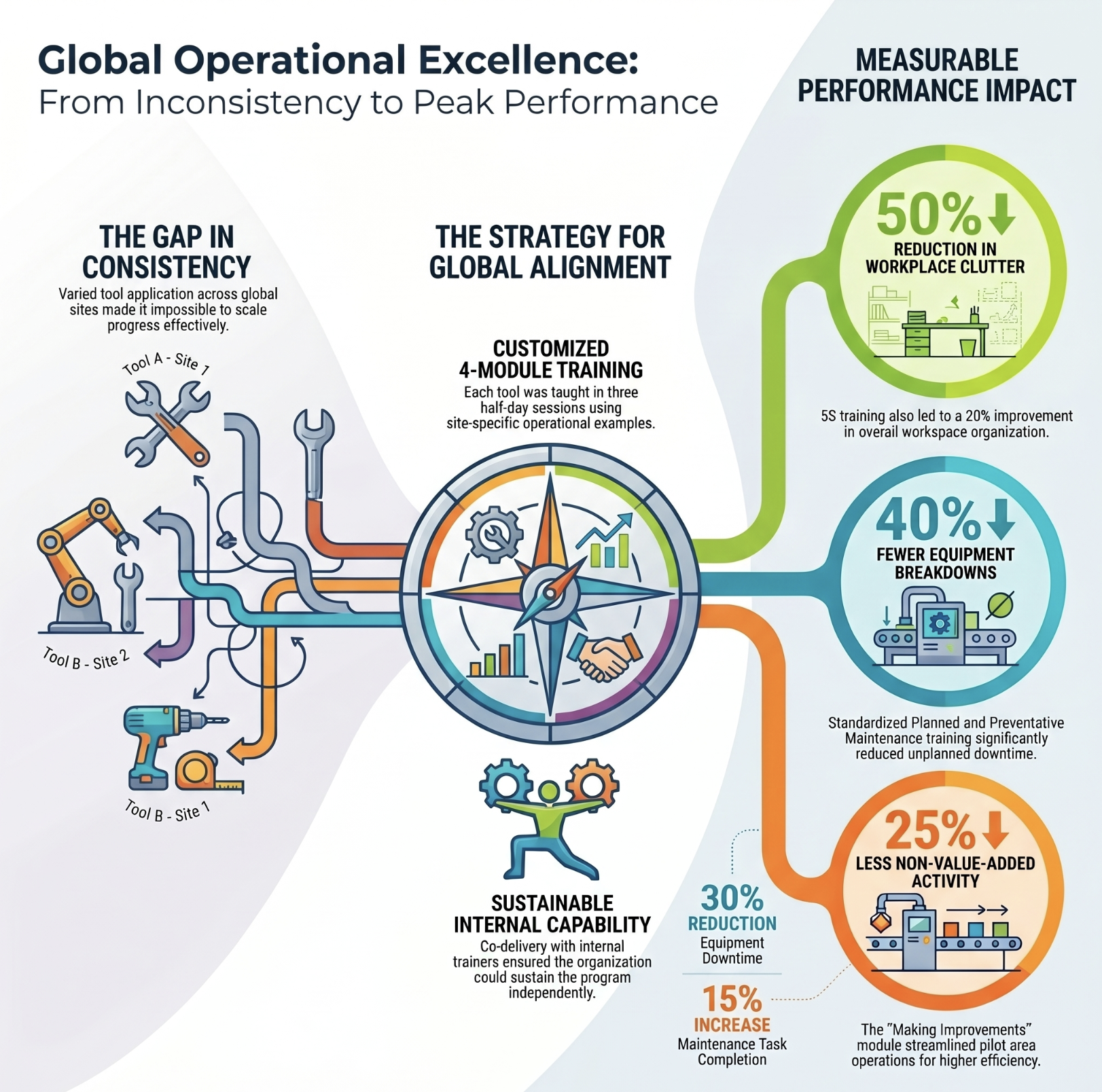
Operational Excellence training standardized across multiple global sites
A global medical device company with four critical OE tools being applied inconsistently across sites — the gap wasn't awareness, it was standardized capability.
Top result: Consistent application of four OE tools across sites. 40% reduction in unplanned equipment breakdowns. Internal trainers equipped to sustain delivery independently.

Product data management harmonized across a global MedTech organization
Fragmented product data processes creating data silos, inconsistent documentation, and workflow inefficiency across multiple functions.
Top result: 35% reduction in data entry errors. Cycle time reduced by 25%. A comprehensive process playbook to sustain the gains.
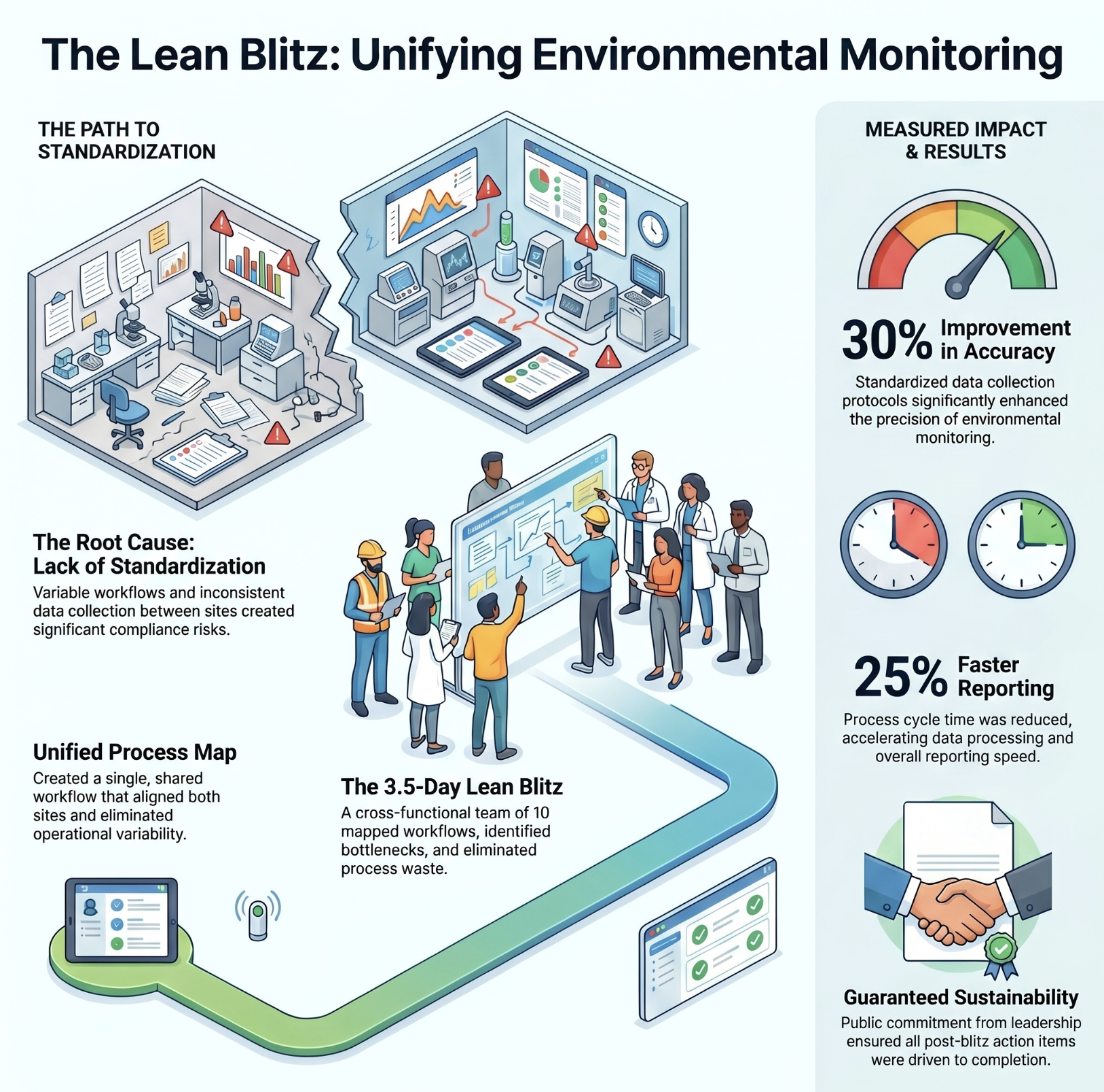
Environmental monitoring standardized across two medical sites
Inconsistencies in Environmental Monitoring workflows causing data collection delays, inefficiencies, and potential compliance risks across two locations.
Top result: Monitoring accuracy improved by 30%. Process cycle time reduced by 25%. A unified process across both sites.
See yourself in any of these stories?
The details change — the industry, the team size, the specific process. But the pattern is almost always the same: capable people blocked by systems that weren't designed to let them succeed. If that sounds familiar, let's talk.
